Chapter 4. Ceramic-on-Polyethylene Bearings in Primary THA
Steven M. Kurtz, PhD.
Jump to a Section
4.1 Introduction
4.2 Wear of Historical and Conventional Polyethylene Against Ceramic Heads.
4.3 Wear of HXLPE Against Ceramic Heads
4.4 Survivorship
4.5 Do Ceramic Bearings Mitigate Taper Corrosion?
4.6 Oxidized Zirconium (Oxinium)
4.7 Summary
4.8 Acknowledgements
4.9 References
4.1 Introduction
In the US, ceramic-on-polyethylene is currently the most widely used alternative to M-PE bearings in THA (Figure 4.1), and in other countries such as the UK, C-PE bearings are increasingly used [1]. As touched upon in previous chapters, laboratory tests of ceramic biomaterials have demonstrated advantages over CoCr for articulation against polyethylene. In vitro, the harder ceramic surfaces have been shown to be more scratch resistant than CoCr femoral heads [2, 3].
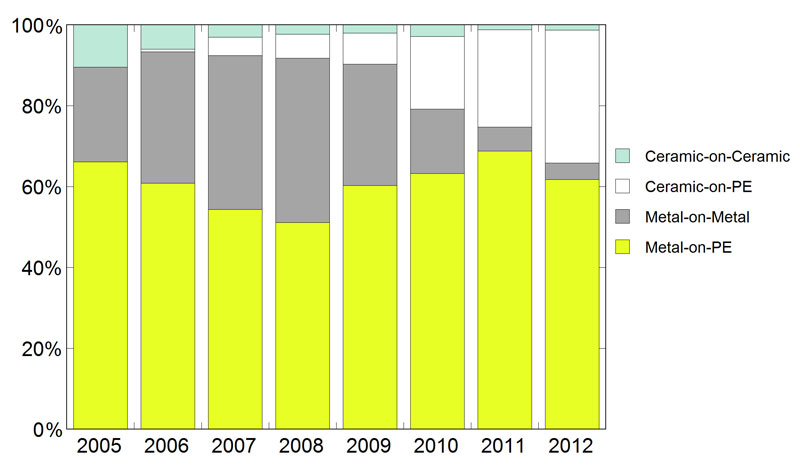
Figure 4.1.Ceramic bearing prevalence for THA estimated from hospital discharge claims records in the United States (2005-2012) [7].
Long-term in vivo studies confirm lower clinical wear rates when ceramic heads are paired with conventional polyethylene [4, 5]. Thus, the widespread acceptance for C-PE articulations in the hip is due to awareness of the long-term reliability in national orthopaedic registries [1, 6], as well as data from the clinical literature [4, 5]. However, there are multiple factors that need to be considered when evaluating clinical studies for contemporary C-PE bearings, including the type of ceramic femoral head and the type of polyethylene counterface (i.e., conventional vs. highly crosslinked polyethylene, HXLPE). For this reason, we discuss the clinical literature for C-PE wear based on the type of polyethylene under consideration.
Most of the papers written about C-PE bearings have focused on wear of polyethylene acetabular liner, because wear was the defining clinical issue of hip replacements up until the 2000s. As a result, the majority of this chapter is devoted to the wear literature as it pertains to C-PE in comparison with M-PE. In the past five years, overall survivorship and taper corrosion have emerged as new clinical metrics for orthopaedic bearings, including C-PE. We have included summaries of these emerging topics after our treatment of clinical wear rates. In this chapter we will focus on synthesizing the literature as it pertains to the outcomes of ceramic-on-polyethylene bearings in primary total hip arthroplasty (THA). We will also explore the published clinical outcomes of oxidized zirconium bearings with polyethylene. If the reader is interested in literature about the use of ceramic bearings in revision surgery, you should skip ahead to Chapter 5.
^ Return to Top
4.2 Wear of Historical and Conventional Polyethylene Against Ceramic Heads
The longest-term clinical studies of C-PE describe its use with conventional polyethylene, which is gas sterilized or gamma sterilized in a low oxygen environment. The body of C-PE literature for conventional polyethylene is still challenging to summarize, because of differences in the study design, ceramic manufacturer, and the type of ceramic investigated. Furthermore, the historical studies of C-PE actually employ gamma-air sterilized polyethylene, as opposed to conventional material.
Sugano et al. [8] reported on the use of first-generation alumina ceramic femoral heads (Bioceram: Kyocera Corporation, Kyoto, Japan) in a series of 57 hips in 50 patients, who were implanted between 1981 and 1983. After 10 years of follow-up, they observed an average wear rate of 0.1 mm/y. Since a control group was not included in this study, the authors did not compare with the wear observed using CoCr heads in a comparable patient population. A similar limitation has been noted in previous retrospective clinical studies reporting wear rates for zirconia against polyethylene, which show, overall an average wear rate of 0.1 mm/y [9]. However, a wear rate of 0.1 mm/y is typically observed in the literature as an average clinical wear rate for CoCr when used with polyethylene [10]. In prospective randomized trials, a significant improvement in wear for zirconia femoral head patients could be detected between CoCr and zirconia femoral heads implanted bilaterally in the same patient for 7.1 years on average [11], whereas a prospective study of two matched cohorts with 4.3 years of follow-up found similar wear rates in both CoCr and zirconia femoral head groups [12]. Other studies have found increased wear associated with zirconia heads compared with stainless steel heads, attributed to phase transformation [13]
Sychterz et al. [14] compared the in vivo wear of a cohort of 81 patients implanted with alumina femoral heads, with a well-matched control group of 43 patients implanted CoCr femoral heads. At seven years follow-up, the radiographic wear rate for patients with alumina was 0.09 ± 0.07 mm/y; the wear rate in the control group was 0.07 ± 0.04 mm/y. The authors concluded that the wear rates of polyethylene in hip replacements using alumina and CoCr femoral heads were similar.
Hendrich et al. [15] reported on the largest matched series of patients with 28-mm diameter alumina (n=100) and CoCr (n=109) femoral heads using the Harris-Galante acetabular component design. The alumina components in this case were identified as BIOLOX (CeramTec). Despite the identical acetabular component designs and femoral head sizes, there were significant differences in follow-up period, which averaged 8y for the CoCr group and 5y for the alumina group. The average wear rates were 0.14±0.11 mm/y for components with CoCr femoral heads, and 0.13±0.08 mm/y for components with the alumina femoral heads. This difference was not statistically significant (p=0.46). Furthermore, a power analysis showed that the clinical study was powered sufficiently to detect a 0.038 mm/y difference (28%) in wear rates with 80% power.
Hopper et al. [16] performed a retrospective analysis of 512 hips (137 ceramic, 430 CoCr heads) that were followed radiographically for femoral head penetration with 3-year minimum follow-up. Due to difficulties encountered in radiographic assessment of zirconia heads, this study focused almost exclusively on alumina femoral heads [17]. 25 factors were examined for their hypothesized influence on wear, including age, gender, preoperative diagnosis, cup design, head diameter, head material, polyethylene material (Enduron vs. Hylamer), liner geometry, polyethylene sterilization method (gas sterilized vs. gamma sterilized). A ceramic femoral head was found to significantly lower the in vivo femoral head penetration rate by 0.03 mm/y, however head material was only the 6th most important factor in the multivariate statistical model, and explained approximately 1% of the variability in the penetration rate data. In comparison, changing the polyethylene material from uncrosslinked, gas sterilized to crosslinked, gamma sterilized was the single most important factor, explaining 12% of the variability in clinical penetration rates.
Urban et al. have also reported long-term clinical performance of alumina C-PE bearings in a retrospective, single-surgeon series with no matching M-PE control group [18]. The 56 patients (64 hips) included in this retrospective study were implanted with a first-generation cemented UHMWPE cup (gamma irradiated in air), a modular 32-mm alumina femoral head, and a stainless steel cemented femoral stem between 1978 and 1981. The patients were originally 69 years old, on average, when they were implanted with a C-PE bearing. At the time of the study, 19 patients with 22 hips were still alive and available for follow-up after a mean implantation time of 18 years (range: 17 to 21 years). The mean penetration rate for this group was 0.034 mm/y in 18 hips with available radiographs (range: 0.000 to 0.077 mm/y), and there was no evidence of osteolysis around any of the acetabular or femoral components. The penetration rates for the C-PE patients surviving long-term implantation were lower than values previously reported for conventional M-PE bearings (typically 0.1 mm/y). However, with only 32% (18/56) of the patients with radiographs available for analysis at the time of the study, the penetration rates for the majority of the cohort could not be reported.
Ihle and colleagues [19] prospectively followed a group of 80 patients implanted with 93 Robert Mathys cups between 1986 and 1989 for 20 years. The polyethylene cups were gamma irradiated in air, and in all cases a 32 mm head was used. 80/93 patients received a BIOLOX® alumina head and 13/93 received a CoCr head. After 19.3 years of follow-up, on average, there no patients lost to follow-up, but 23 patients (with 26 hips) died prior to the completion of the study. Of the 47 alumina cases and 8 CoCr cases available for analysis, the average (±SD) linear penetration rate in the ceramic cohort (0.107±0.065 mm/y) was significantly lower than in the metal cohort (0.190±0.124 mm/y, p = 0.025). The authors also noted significant higher penetration rates for those patients who were revised during the study period (0.191 mm/y), as compared to those that were not (0.105 mm/y, p≤0.005).
Two recent clinical studies [4, 5] have employed matched cohort clinical study designs to compare wear rates for bilateral C-PE vs. M-PE using conventional polyethylene. Wang et al. [5] evaluated 22 patients (11 men and 11 women) who were implanted in 2002 with ENDURON (a trade name for DePuy Synthes, denoting conventional polyethylene). The patients received 28 mm BIOLOX®forte heads in one hip, and 28 mm CoCr heads on the contralateral side. Radiographic linear penetration was measured using the Dorr method. On the C-PE side, the average penetration rate (±SD) was 0.056 ± 0.032 mm/y, and significantly less than on the M-PE side, which was 0.133 ± 0.045 mm/y (p < 0.001). There was one M-PE hip revised, but not in the C-PE side.
Meftah and colleagues [4] studied two matched cohorts of patients who were implanted with either 28 mm BIOLOX® alumina or CoCr femoral heads between 1989 and 1991. The acetabular liners were fabricated from GUR 4150 and sterilized in argon gas. Average followup was 17 ± 1.2 years. Radiographic linear penetration was measured using a computerized version of the Livermore technique. On the C-PE side, the average penetration rate (±SD) was 0.086 ± 0.05 mm/y, and significantly less than on the M-PE side, which was 0.137 ± 0.05 mm/y (p = 0.0015). Three of the M-PE hips were revised due to osteolysis, but none in the C-PE cohort.
In summary, the clinical history for ceramic-on-conventional polyethylene in the literature includes both alumina and zirconia femoral heads. It thus remains difficult to generalize about C-PE bearings, because the clinical performance over the years has been ceramic-material specific, especially when zirconia is concerned. Many of the older studies employed gamma-air, historical polyethylene. Notwithstanding these limitations, several long-term clinical studies have now demonstrated lower wear rates for conventional polyethylene when comparing alumina with CoCr femoral heads at 10 years or greater followup [4, 5].
^ Return to Top
4.3 Wear of HXLPE Against Ceramic Heads
The clinical literature for ceramic articulations with HXLPE is as complex and nuanced as the studies dealing with conventional or historical polyethylene. Perhaps the oldest study of crosslinked polyethylene with ceramic heads was performed by Wroblewski et al. from Wrightington hospital, who performed a long-term prospective observational cohort study on a group of 17 patients implanted with 19 experimental, silane-crosslinked HDPE acetabular cups and 22 mm diameter alumina femoral heads [20, 21]. These patients were relatively young, with an average age of 47 years (range: 26 to 58 years) at the beginning of the study. The cemented cups were similar in design to the Low Friction Arthroplasty developed by Charnley [22]. The most recent follow-up of this cohort after an average of 17 years of implantation (range: 15 to 18.1 years) was completed for 9 patients with 11 hips [21]. The clinical results for this group were judged to be excellent, and the mean radiographic penetration rate was 0.019 mm/y. None of the patients showed evidence of osteolysis on radiographs. Although the young patients in this study showed evidence of much lower wear than patients implanted with conventional M-PE bearings at this institution, it is unclear whether the wear reduction can be attributed to the novel chemically crosslinked acetabular cups, the alumina femoral heads, or the C-PE bearing combination.
Numerous studies investigating the wear behavior of commercially available, ceramic-on-HXLPE have been reported in the literature (Table 4.1). The two-dimensional ceramic femoral head penetration rate articulating against HXLPE has been reported to range between 0.00 and 0.10 mm/y (Table 4.1), depending upon the type of HXLPE, the type of ceramic, the radiographic wear measurement technique, and the study design. An RCT study by Kim et al. [23] reported a two-dimensional linear penetration rate for alumina on HXLPE to be 0.06 mm/year (SE, 0.003). Similarly, Oonishi et al. [24] reported a penetration rate of 0.10 mm/y (SE, 0.006) for alumina on HXLPE. Nakahara and coworkers [25] compared steady-state penetration rates for Longevity paired with a zirconia femoral head. Two RCTs have reported penetration rate data for zirconia femoral heads articulating with Aeonian (Kyocera/JMM) [26, 27](Table 4.1).
Table 4.1.Summary of Linear Penetration Reported for Ceramic-on-HXLPE studies
| Ceramic head material (manufacturer) | Mean (mm/year; standard error) | HXLPE liner | Study | Study type |
| Zirconia (PHS head, Kyocera Corp) | 0.067 (0.009) | Aeonian | Ise 2009 [26] | RCT |
| Zirconia (HHZ head, Kobelco, Kobe Steel Ltd) | 0.059 (0.006) | Aeonian | Ise 2009 [26] | RCT |
| Zirconia (3Y-TZP, Japan Medical Material) | 0.000 (0.005) | Aeonian | Kawate 2009 [27] | RCT |
| Zirconia (NGK Spark Plug) | 0.030 (0.002) | Longevity | Nakahara 2009 [25] | COH |
| Alumina (Kyocera Medical) | 0.100 (0.006) | Aeonian | Oonishi 2006 [24] | COH |
| Zirconia-toughened Alumina (ZTA) (BIOLOX®delta, CeramTec) | 0.006 (0.014) | X3 | Meftah 2011 [28] | COH |
| Alumina (BIOLOX®forte, CeramTec) | 0.031 (0.004) | Marathon | Kim 2013 [29] | RCT |
| Alumina (BIOLOX®forte, CeramTec) | 0.019 (0.013) | Crossfire | Epinette 2014 [30] | COH |
^ Return to Top
4.4 Survivorship
The survivorship of ceramic-on-HXLPE documented in the literature has recently been comprehensively studied in a systemic review and network meta-analysis by Wyles and colleagues [31]. They found similar short- to intermediate- survivorship when comparing ceramic-on-HXLPE to metal-on-HXLPE in Level I randomized controlled trials (RCTs) with patients younger than 65 years in age. However, the 18 RCTs they drew upon “highlight important deficits within the current standard of orthopaedic evidence” [31], due to small sample sizes, loss to follow up, and short- to intermediate-term surveillance periods.
We must turn to orthopaedic registries for comparisons of long-term survivorship of ceramic-on-HXLPE using contemporary implant designs. Starting with the 2014 Annual Report [6], the Australian Registry has begun reporting data by type of ceramic head articulating against conventional or HXLPE. The most recent registry data shows similar survivorship when comparing different ceramics against HXLPE for femoral head diameters less than or equal to 28 mm. The registry also reports similar overall survivorship for C-HXLPE and M-HXLPE. Specifically, in 2014 Australian registry reports 4.5% cumulative risk of revision at 10 years for ceramic-on-HXLPE bearings (95% confidence intervals: 3.9-5.1%), as compared with 4.3% (95% CI: 4.1-4.5%) for metal-on-HXLPE bearings (see Table HT21 in [6]). These data include all head sizes, methods of fixation, and are specific to monolithic stems without an exchangeable or modular femoral neck.
The National Joint Registry of England and Wales (NJR) is somewhat less detailed in terms of its classification of polyethylene and ceramic biomaterials, so that only granularity that was available as of the 2014 annual report was based on cement fixation [1]. Specifically, in 2014 NJR reports 2.1% cumulative risk of revision at 10 years for cemented C-PE bearings (95% CI: 1.7-2.5%), as compared with 3.1% (95% CI: 3.0-3.3%) for cemented M-PE bearings (see Table 3.7 in [1]). For uncemented primary THA, the 10-year revision risk was reported to be 3.7% (95% CI: 3.2-4.3%) for C-PE as compared with 4.0% (95% CI: 3.7-4.3%) for M-PE (see Table 3.7 in [1]). The 2014 NJR considered cemented C-PE total hips to be the “most ‘successful’ bearing combination” based on its survivorship.
The registry data, while encouraging, must also be interpreted with caution due to an entirely different set of inherent limitations. In an “all comers” registry study it is difficult to properly adjust for differences in patient, clinical, and surgical factors, all of which may influence the long-term survival of the total hip surgical procedure. It is clear, however, the increasing importance of this type of research, which is still continuing today. Due to the importance of this topic we expect registries to adopt more sophisticated methods for analyzing their data in the future, in order to hopefully provide a stronger comparison based on the available evidence.
^ Return to Top
4.5 Do Ceramic Bearings Mitigate Taper Corrosion?
Ceramics have recently been introduced as a solution for metal release caused by taper corrosion between the femoral head and the stem trunnion [32]. As a research topic, taper corrosion is not a new issue in orthopedics. The metallic biomaterials we use in hip and knee implants, namely cobalt and titanium based alloys, are among the most corrosion resistant materials for implant use and have a clinical track record spanning many decades. In the 1980s and 1990s, researchers studied in detail corrosion at the modular head-stem junction [33-38]. These early studies helped identify the corrosion products from modular connections in metallic stems, as well as establish the corrosion mechanism, which is now recognized to be a complex mechanically assisted crevice corrosion process. However, all of this basic science, mechanistic research was almost exclusively based on cobalt chrome alloy femoral heads on either cobalt-based or titanium-based alloy femoral stems.
With the growing concern about taper corrosion in modular large-head, metal-on-metal hip designs [39, 40]; two-piece, modular femoral stems [41]; and, most recently, metal-on-polyethylene hip designs [42], there is increased awareness of the issue of metal debris release in contemporary modular connections and the potential for rare complications, including adverse local tissue reactions (ALTRs). Until recently, the literature has been largely silent on the topic of taper corrosion used in conjunction with ceramic femoral heads. In this section we briefly summarize some of the key studies examining taper corrosion with ceramic femoral heads in modular hip systems, and share an evolving perspective on this topic.
When surveying the issue of taper corrosion products in the 1990s, researchers including Urban, Gilbert, Jacobs and colleagues studied a broad range of hip implant designs, with an emphasis on modular connections in which both the head and stem tapers were metallic [35-38, 43, 44]. However, one of the samples in their collection was an Autophor hip, an early cobalt-chromium alloy stem with an alumina femoral head, fabricated from first-generation BIOLOX ceramic. Based on this single ceramic retrieval, and their larger collection of metallic components, Urban and colleagues [37] observed that, “the products of corrosion identified at the modular junctions of all of the various prostheses examined were similar regardless of the implant design or materials coupled, even when a ceramic head was employed.” After this early research established the basic mechanism of taper corrosion, the issue of modular taper corrosion largely faded from the forefront of orthopaedic consciousness, replaced by the problem of osteolysis and aseptic loosening due to polyethylene wear which dominated orthopaedic meetings in the 1990s.
In 2004, Hallab and colleagues from Rush University published an in vitro study explicitly directed at comparing taper corrosion using ceramic heads with cobalt chrome alloy heads [45]. The authors theorized that fretting would be more severe between a ceramic head and metal stem, and hypothesized that ceramic head systems would release more debris than metal heads. To test their hypothesis, they selected CoCr heads and CoCr stems by a single manufacturer. The ceramic heads were zirconia, manufactured by a supplier that has since withdrawn from the orthopaedic market. Unexpectedly, the researchers found that the CoCr stems with CoCr heads released 11 times greater Co and 3 times greater Cr than the stems fitted with zirconia heads. Although the research findings refuted their original hypothesis, the authors cautioned against overgeneralizing their results to other designs and implant systems, and concluded that “ultimately, it will be through careful evaluation of clinical performance and implant retrieval analysis that the relative performance of ceramic-metal modular junctions will be ascertained” [45].
More recently, researchers from Drexel University carefully matched retrieved implants in the two 50-implant cohorts (100 patients total) to account for all the variables influencing taper corrosion and to focus on the difference between ceramic-metal and metal-metal tapers [32]. All of the hip implants in the ceramic head cohort were from ceramic-on-polyethylene or ceramic-on-ceramic bearings. The hip implants from the metal head cohort were all from metal-on-polyethylene bearings. The stems included a variety of cobalt and titanium based alloys from the major manufacturers, and both the stem design and offset were matched between the ceramic and metal head cohorts. The ceramic heads were all BIOLOX alumina or delta heads produced by CeramTec. The stem tapers in each cohort were scored from 1 (mild) to 4 (severe) by three independent observers for evidence of fretting and corrosion using a scale adapted by Goldberg and associates for retrieved metal tapers [46]. The researchers observed significantly less corrosion among the stems in the ceramic-head cohort as compared with the metal head cohort (Figure 4.2). The median fretting and corrosion score was 2 for the metal stems mated to a ceramic head, as compared to the metal stems mated with a CoCr head, which had a median score of 3 (Figure 4.2).
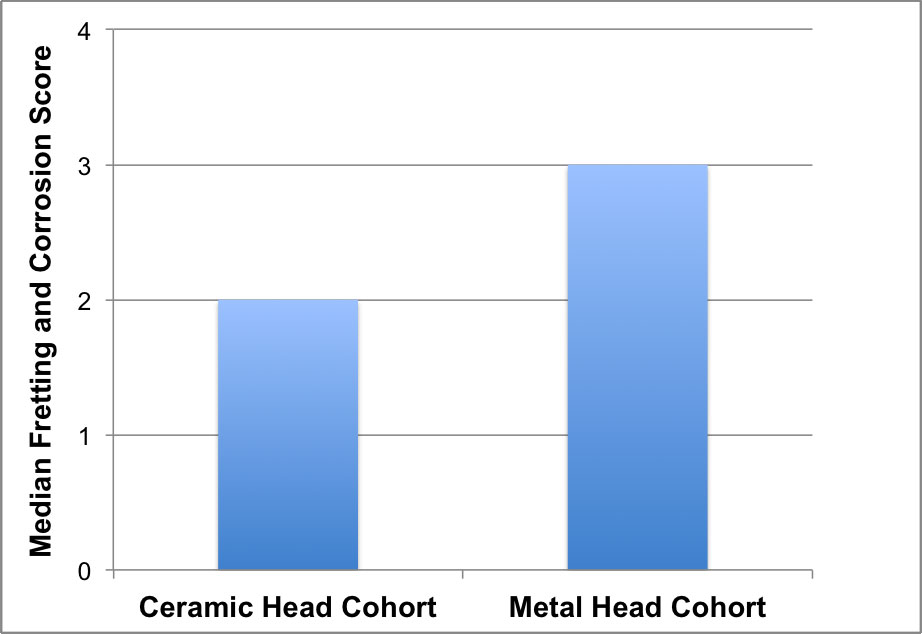
Figure 4.2.The retrieved stem tapers mated with CoCr alloy femoral heads exhibited significantly less taper corrosion as compared with the stems with ceramic heads. Adapted from [32].
In a second study, the Drexel researchers developed and validated a highly sensitive technique to measure the microscopic volumes of metal that are released in CoCr femoral heads and metal stem tapers [47]. After re-examining the previous matched 50-50 cohort of ceramic and femoral heads, researchers found that the ceramic cohort exhibited over 90% less metal release, in terms of volumetric material loss, when compared with the CoCr cohort (Figure 4.3). When focusing on the CoCr head cohort, they found that the vast majority of the metal release, again, over 90%, was liberated from the femoral head, rather than the stem taper.
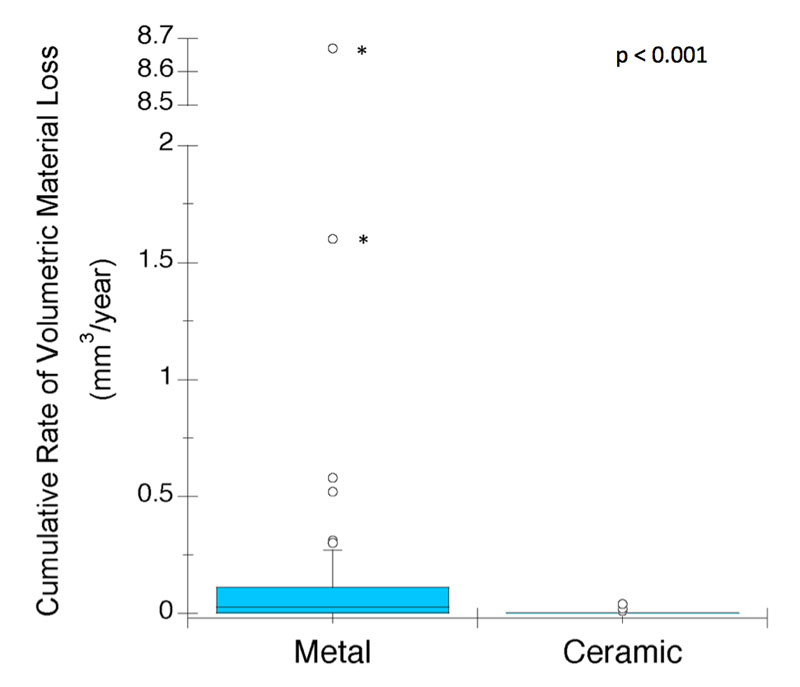
Figure 4.3.The modular tapers with CoCr alloy femoral heads exhibited significantly greater volumetric material loss as compared with the stems with ceramic heads. Adapted from [47].
^ Return to Top
4.6 Oxidized Zirconium (Oxinium)
As discussed in Chapter 2, oxidized zirconium femoral heads (Oxinium, Smith and Nephew) have been clinically used since 1997 as a femoral head material in articulations first with conventional polyethylene and, more recently, with HXLPE. Concerns have been raised in the literature that the 4-micron, thin layer of zirconia ceramic on the surface of these metal heads may be susceptible to damage in vivo. Starting in 2007, a number of case studies [75-81] for dislocated femoral heads demonstrated severe scratching and excavation of the zirconia surface layer, exposing the softer metallic zirconium alloy beneath. An example of this type of in vivo damage of oxidized zirconium is shown in Figure 4.4. The clinical implications underlying these case studies was that closed reduction, which is normally performed as the preferred initial treatment for dislocation, may cause severe damage to the Oxinium femoral head and require replacement during revision to prevent long-term wear of the polyethylene liner.

Figure 4.4.Example of surface damage to a retrieved oxidized zirconium femoral head. This component was revised due to recurring dislocation after 3 years in vivo. Images courtesy of Sevi Koçagoz, Drexel University.
Moussa and colleagues [82] from the Hospital for Special Surgery in New York have performed the largest retrieval study to date looking at in vivo changes in retrieved oxidized zirconium femoral heads. In a collection of 59 retrievals, they found that the revision reason was a predictor of the severe type of damage shown as an example in Figure 4.4. Among the 24 heads revised due to dislocation, 75% were severely damaged, whereas only 14% of the heads revised for reasons other than dislocation were severely damaged. Like previous investigators, Moussa et al. found that the explanted oxidized zirconium heads had a significantly higher surface roughness in severely damaged regions. They used SEM with EDAX to confirm that the damaged regions on the retrieved heads were due to effacement of the oxidized zirconium layer with exposure of the metal underneath, as opposed to metal transfer on the surface of the head. It must be emphasized that Moussa’s femoral heads were all obtained at revision, and therefore do not necessarily reflect the disposition of well-functioning heads in vivo.
Clinical studies have been published for Oxinium in THA with both two-year [83, 84] follow-up and, most recently, with five-year follow-up [85-88]. The intermediate term studies emphasized the importance of monitoring patients for evidence of accelerated wear that could be attributed to the severe types of surface damage shown in Figure 3.4. To date, none of the available studies show evidence that the severe damage observed in some explanted components at revision surgery is predictive of the clinical performance in well-functioning implants. Of note, the randomized trial by Jassim [87] compared three cohorts: Oxinium/HXLPE, CoCr/HXLPE, and Oxinium/polyethylene. They found that the use of HXLPE was more important than the choice of CoCr or Oxinium for reducing wear. Furthermore, two patients with recurring dislocation and closed reduction, who received an oxidized zirconium head and HXLPE liner, did not exhibit elevated wear rates when compared with other patients in their cohort. Similar findings were echoed in the randomized studies by Zaoui [88] and Morison [85].
It may be that longer-term studies are needed to determine if Oxinium has measurable clinical benefits over CoCr femoral heads when paired with an HXLPE liner. At present, Oxinium’s vulnerability to damage caused by dislocation and closed reduction has not been shown to have a negative clinical effect in well-functioning hip implants.
^ Return to Top
4.7 Summary
Historically, ceramic heads have been studied almost exclusively in terms of their ability to reduce wear at the articulating surface. Indeed, as we have seen in this chapter, many studies have shown evidence of reduced wear with ceramic heads against conventional polyethylene, although it remains difficult to quantify the reduced wear provided by ceramic heads when paired with HXLPE. Beyond wear reduction, recent research indicates that by using a ceramic femoral head, Co and Cr fretting and corrosion from the modular head-neck taper is mitigated. Thus, the evidence is clear that, in addition to wear reduction, ceramic heads are a practical and effective solution for mitigating taper corrosion.
C-PE bearings have outstanding clinical performance as documented in the international registries, comparable to COC. In previous decades, prior to the development of HXLPE, the distinction between COC and C-PE may have been relatively straightforward. However, with the advent of modern HXLPE materials and contemporary implant designs, the differences in clinical performance between C-PE and COC are less easily discernable [31, 90, 91]. As we have seen throughout this chapter, the clinical and registry studies with 10-year follow up show comparable, excellent survivorship with both types of ceramic bearings. For these reasons, ceramic bearing selection directly comparing C-PE with COC will continue to be an important topic debated in the literature for the foreseeable future.
4.8 Acknowledgements
The author would like to thank Christina Arnholt, Drexel University, for her editorial assistance with this chapter.
^ Return to Top
4.9 References for Chapter 4
[1] National Joint Registry for England and Wales. 11th Annual NJR Report. London: NJR; 2014. [2] Lancaster JG, Dowson D, Isaac GH, Fisher J. The wear of ultra-high molecular weight polyethylene sliding on metallic and ceramic counterfaces representative of current femoral surfaces in joint replacement. Proceedings of the Institution of Mechanical Engineers. 1997;211:17-24. [3] Cuckler JM, Bearcroft J, Asgian CM. Femoral head technologies to reduce polyethylene wear in total hip arthroplasty. Clin Orthop. 1995;317:57-63. [4] Meftah M, Klingenstein GG, Yun RJ, Ranawat AS, Ranawat CS. Long-term performance of ceramic and metal femoral heads on conventional polyethylene in young and active patients: a matched-pair analysis. J Bone Joint Surg Am. 2013;95:1193-7. [5] Wang S, Zhang S, Zhao Y. A comparison of polyethylene wear between cobalt-chrome ball heads and alumina ball heads after total hip arthroplasty: a 10-year follow-up. J Orthop Surg Res. 2013;8:20. [6] Australian Orthopaedic Association National Joint Replacement Registry. Annual Report. Adelaide: AOA; 2014. [7] Agency for Healthcare Research and Quality. Nationwide Inpatient Sample. [8] Sugano N, Nishii T, Nakata K, Masuhara K, Takaoka K. Polyethylene sockets and alumina ceramic heads in cemented total hip arthroplasty. A ten-year study. J Bone Joint Surg Br. 1995;77:548-56. [9] Cales B. Zirconia as a sliding material: histologic, laboratory, and clinical data. Clin Orthop. 2000;379:94-112. [10] Dumbleton JH, Manley MT, Edidin AA. A literature review of the association between wear rate and osteolysis in total hip arthroplasty. J Arthroplasty. 2002;17:649-61. [11] Kim YH. Comparison of polyethylene wear associated with cobalt-chromium and zirconia heads after total hip replacement. A prospective, randomized study. J Bone Joint Surg Am. 2005;87:1769-76. [12] Kraay MJ, Thomas RD, Rimnac CM, Fitzgerald SJ, Goldberg VM. Zirconia versus Co-Cr femoral heads in total hip arthroplasty: early assessment of wear. Clin Orthop Relat Res. 2006;453:86-90. [13] Hernigou P, Bahrami T. Zirconia and alumina ceramics in comparison with stainless-steel heads. Polyethylene wear after a minimum ten-year follow-up. J Bone Joint Surg Br. 2003;85:504-9. [14] Sychterz CJ, Engh CA, Jr., Young AM, Hopper RH, Jr., Engh CA. Comparison of in vivo wear between polyethylene liners articulating with ceramic and cobalt-chrome femoral heads. J Bone Joint Surg Br. 2000;82:948-51. [15] Hendrich C, Goebel S, Roller C, Kirschner S, Martell JM. Wear performance of 28 millimeter femoral heads with the Harris-Galante cup: Comparison of alumina and cobalt chrome. In: Zippel H, Dietrich M, editors. Bioceramics in Joint Arthroplasty, 8th Biolox Symposium Proceedings. Darmstadt: Steinkopff Verlag; 2003. p. 177-82. [16] Hopper RH, Jr., Engh CA, Jr., Fowlkes LB, Engh CA. The pros and cons of polyethylene sterilization with gamma irradiation. Clin Orthop Relat Res. 2004:54-62. [17] Hopper RH, Jr. Personal communication. AORI; 2008. [18] Urban JA, Garvin KL, Boese CK, Bryson L, Pedersen DR, Callaghan JJ, Miller RK. Ceramic-on-polyethylene bearing surfaces in total hip arthroplasty. Seventeen to twenty-one-year results. J Bone Joint Surg Am. 2001;83-a:1688-94. [19] Ihle M, Mai S, Siebert W. Ceramic versus metal femoral heads in combination with polyethylene cups: Long-term wear analysis at 20 years. Semin Ortho. 2011;22:218-24. [20] Wroblewski BM, Siney PD, Dowson D, Collins SN. Prospective clinical and joint simulator studies of a new total hip arthroplasty using alumina ceramic heads and cross-linked polyethylene cups. Journal of Bone & Joint Surgery. 1996;78 B:280-5. [21] Wroblewski BM, Siney PD, Fleming PA. Low-friction arthroplasty of the hip using alumina ceramic and cross-linked polyethylene. A 17-year follow-up report. J Bone Joint Surg Br. 2005;87:1220-1. [22] Charnley J. Total hip replacement by low-friction arthroplasty. Clin Orthop. 1970;72:7-21. [23] Kim YH, Kim JS, Choi YW, Kwon OR. Intermediate results of simultaneous alumina-on-alumina bearing and alumina-on-highly cross-linked polyethylene bearing total hip arthroplasties. J Arthroplasty. 2009;24:885-91. [24] Oonishi H, Kim SC, Takao Y, Kyomoto M, Iwamoto M, Ueno M. Wear of highly cross-linked polyethylene acetabular cup in Japan. The Journal of arthroplasty. 2006;21:944-9. [25] Nakahara I, Nakamura N, Nishii T, Miki H, Sakai T, Sugano N. Minimum Five-Year Follow-Up Wear Measurement of Longevity Highly Cross-Linked Polyethylene Cup Against Cobalt-Chromium or Zirconia Heads. J Arthroplasty. 2009. [26] Ise K, Kawanabe K, Tamura J, Akiyama H, Goto K, Nakamura T. Clinical results of the wear performance of cross-linked polyethylene in total hip arthroplasty: prospective randomized trial. The Journal of arthroplasty. 2009;24:1216-20. [27] Kawate K, Ohmura T, Kawahara I, Tamai K, Ueha T, Takemura K. Differences in highly cross-linked polyethylene wear between zirconia and cobalt-chromium femoral heads in Japanese patients: a prospective, randomized study. The Journal of arthroplasty. 2009;24:1221-4. [28] Meftah M, Ebrahimpour PB, He C, Ranawat AS, Ranawat CS. Preliminary clinical and radiographic results of large ceramic heads on highly cross-linked polyethylene. Orthopedics. 2011;34:133. [29] Kim YH, Park JW, Kulkarni SS, Kim YH. A randomised prospective evaluation of ceramic-on-ceramic and ceramic-on-highly cross-linked polyethylene bearings in the same patients with primary cementless total hip arthroplasty. International orthopaedics. 2013;37:2131-7. [30] Epinette JA, Manley MT. No differences found in bearing related hip survivorship at 10-12 years follow-up between patients with ceramic on highly cross-linked polyethylene bearings compared to patients with ceramic on ceramic bearings. J Arthroplasty. 2014;29:1369-72. [31] Wyles CC, Jimenez-Almonte JH, Murad MH, Norambuena-Morales GA, Cabanela ME, Sierra RJ, Trousdale RT. There Are No Differences in Short- to Mid-term Survivorship Among Total Hip-bearing Surface Options: A Network Meta-analysis. Clin Orthop Relat Res. 2015;473:2031-41. [32] Kurtz SM, Kocagoz SB, Hanzlik JA, Underwood RJ, Gilbert JL, Macdonald DW, Lee GC, Mont MA, Kraay MJ, Klein GR, Parvizi J, Rimnac CM. Do ceramic femoral heads reduce taper fretting corrosion in hip arthroplasty? A retrieval study. Clin Orthop Relat Res. 2013;471:3270-82. [33] Black J. Does Corrosion Matter? The Journal of Bone & Joint Surgery1988. p. 517-20. [34] Collier JP, Surprenant VA, Jensen RE, Mayor MB. Corrosion at the interface of cobalt-alloy heads on titanium-alloy stems. Clin Orthop Relat Res. 1991:305-12. [35] Gilbert JL, Buckley CA, Jacobs JJ. In vivo corrosion of modular hip prosthesis components in mixed and similar metal combinations. The effect of crevice, stress, motion, and alloy coupling. J Biomed Mater Res. 1993;27:1533-44. [36] Gilbert JL, Jacobs JJ. The mechanical and electrochemical processes associated with taper fretting crevice corrosion: a review. In: Marlowe D, Parr J, Mayor MB, editors. Modularity of Orthopedic Implants. Conshohocken, PA: ASTM; 1997. p. 45-59. [37] Urban RM, Jacobs JJ, Gilbert JL, Rice SB, Jasty M, Bragdon CR, Galante JO. Characterization of solid products of corrosion generated by modular-head femoral stems of different designs and materials, STP 1301. In: Marlowe D, Parr J, Mayor MB, editors. Modularity of Orthopedic Implants. Conshohocken, PA: ASTM; 1997. p. 33-44. [38] Jacobs JJ, Gilbert JL, Urban RM. Corrosion of metal orthopaedic implants. J Bone Joint Surg Am. 1998;80:268-82. [39] Langton DJ, Sidaginamale R, Lord JK, Nargol AV, Joyce TJ. Taper junction failure in large-diameter metal-on-metal bearings. Bone & Joint Research - Hip. 2012;1:56-63. [40] Langton DJ, Jameson SS, Joyce TJ, Gandhi JN, Sidaginamale R, Mereddy P, Lord J, Nargol AV. Accelerating failure rate of the ASR total hip replacement. J Bone Joint Surg Br. 2011;93:1011-6. [41] Gill IP, Webb J, Sloan K, Beaver RJ. Corrosion at the neck-stem junction as a cause of metal ion release and pseudotumour formation. J Bone Joint Surg Br. 2012;94:895-900. [42] Cooper HJ, Della Valle C, Berger RA, Tetreault BA, Paprosky W, Sporer S, Jacobs JJ. Corrosion at the Head-Neck Taper as a Cause for Adverse Local Tissue Reactions After Total Hip Arthroplasty. Journal of Bone and Joint Surgery, Am. 2012;94:1655 - 61. [43] Urban RM, Jacobs JJ, Gilbert JL, Galante JO. Migration of corrosion products from modular hip prostheses. Particle microanalysis and histopathological findings. J Bone Joint Surg Am. 1994;76:1345-59. [44] Jacobs JJ, Urban RM, Gilbert JL, Skipor AK, Black J, Jasty M, Galante JO. Local and distant products from modularity. Clin Orthop Relat Res. 1995:94-105. [45] Hallab NJ, Messina C, Skipor A, Jacobs JJ. Differences in the fretting corrosion of metal-metal and ceramic-metal modular junctions of total hip replacements. J Orthop Res. 2004;22:250-9. [46] Goldberg JR, Gilbert JL, Jacobs JJ, Bauer TW, Paprosky W, Leurgans S. A multicenter retrieval study of the taper interfaces of modular hip prostheses. Clin Orthop Relat Res. 2002:149-61. [47] Kocagoz SB, Underwood RJ, MacDonald D, Gilbert JL, Rimnac CM, Kurtz SM. Metal release in ceramic and CoCr heads at the modular junction: a matched cohort retrieval study. Trans Orthop Res Soc. 2015;Poster Session PS1-065:0887. [48] Jeffers JR, Walter WL. Ceramic-on-ceramic bearings in hip arthroplasty: state of the art and the future. J Bone Joint Surg Br. 2012;94:735-45. [49] Vendittoli PA, Riviere C, Lavigne M, Lavoie P, Alghamdi A, Duval N. Alumina on alumina versus metal on conventional polyethylene: a randomized clinical trial with 9 to 15 years follow-up. Acta orthopaedica Belgica. 2013;79:181-90. [50] Boyer P, Huten D, Loriaut P, Lestrat V, Jeanrot C, Massin P. Is alumina-on-alumina ceramic bearings total hip replacement the right choice in patients younger than 50 years of age? A 7- to 15-year follow-up study. Orthopaedics & traumatology, surgery & research : OTSR. 2010;96:616-22. [51] Kim YH, Choi Y, Kim JS. Cementless total hip arthroplasty with ceramic-on-ceramic bearing in patients younger than 45 years with femoral-head osteonecrosis. International orthopaedics. 2010;34:1123-7. [52] Kress AM, Schmidt R, Holzwarth U, Forst R, Mueller LA. Excellent results with cementless total hip arthroplasty and alumina-on-alumina pairing: minimum ten-year follow-up. International orthopaedics. 2011;35:195-200. [53] Lee YK, Ha YC, Yoo JJ, Koo KH, Yoon KS, Kim HJ. Alumina-on-alumina total hip arthroplasty: a concise follow-up, at a minimum of ten years, of a previous report. The Journal of bone and joint surgery American volume. 2010;92:1715-9. [54] D'Antonio JA, Capello WN, Naughton M. Ceramic bearings for total hip arthroplasty have high survivorship at 10 years. Clin Orthop Relat Res. 2012;470:373-81. [55] D'Antonio JA, Capello WN, Naughton M. High survivorship with a titanium-encased alumina ceramic bearing for total hip arthroplasty. Clin Orthop Relat Res. 2014;472:611-6. [56] Sugano N, Takao M, Sakai T, Nishii T, Miki H, Ohzono K. Eleven- to 14-year follow-up results of cementless total hip arthroplasty using a third-generation alumina ceramic-on-ceramic bearing. J Arthroplasty. 2012;27:736-41. [57] Solarino G, Piazzolla A, Notarnicola A, Moretti L, Tafuri S, De Giorgi S, Moretti B. Long-term results of 32-mm alumina-on-alumina THA for avascular necrosis of the femoral head. J Orthop Traumatol. 2012;13:21-7. [58] Kang BJ, Ha YC, Ham DW, Hwang SC, Lee YK, Koo KH. Third-generation alumina-on-alumina total hip arthroplasty: 14 to 16-year follow-up study. J Arthroplasty. 2015;30:411-5. [59] Hamilton WG, McAuley JP, Blumenfeld TJ, Lesko JP, Himden SE, Dennis DA. Midterm Results of Delta Ceramic-on-Ceramic Total Hip Arthroplasty. J Arthroplasty. 2015. [60] Cai P, Hu Y, Xie J. Large-diameter Delta ceramic-on-ceramic versus common-sized ceramic-on-polyethylene bearings in THA. Orthopedics. 2012;35:e1307-13. [61] Baek SH, Kim WK, Kim JY, Kim SY. Do Alumina Matrix Composite Bearings Decrease Hip Noises and Bearing Fractures at a Minimum of 5 Years After THA? Clin Orthop Relat Res. 2015. [62] Shin YS, Han SB, Jung TW. Comparison between preassembled and modular cups in primary cementless total hip arthroplasty: a two-year minimum follow-up study. J Arthroplasty. 2014;29:2378-82. [63] Aoude AA, Antoniou J, Epure LM, Huk OL, Zukor DJ, Tanzer M. Midterm Outcomes of the Recently FDA Approved Ceramic on Ceramic Bearing in Total Hip Arthroplasty Patients Under 65Years of Age. J Arthroplasty. 2015;30:1388-92. [64] Wang W, Guo W, Yue D, Shi Z, Zhang N, Liu Z, Sun W, Wang B, Li Z. Fourth-generation ceramic-on-ceramic total hip arthroplasty in patients of 55 years or younger: short-term results and complications analysis. Chin Med J (Engl). 2014;127:2310-5. [65] McDonnell SM, Boyce G, Bare J, Young D, Shimmin AJ. The incidence of noise generation arising from the large-diameter Delta Motion ceramic total hip bearing. The bone & joint journal. 2013;95-B:160-5. [66] Tai SM, Munir S, Walter WL, Pearce SJ, Walter WK, Zicat BA. Squeaking in Large Diameter Ceramic-on-Ceramic Bearings in Total Hip Arthroplasty. J Arthroplasty. 2015;30:282-5. [67] Colwell CW, Jr., Hozack WJ, Mesko JW, D'Antonio JA, Bierbaum BE, Capello WN, Jaffe WL, Mai KT. Ceramic-on-ceramic total hip arthroplasty early dislocation rate. Clin Orthop Relat Res. 2007;465:155-8. [68] Mai K, Hardwick ME, Walker RH, Copp SN, Ezzet KA, Colwell CW, Jr. Early dislocation rate in ceramic-on-ceramic total hip arthroplasty. HSS J. 2008;4:10-3. [69] Hernigou P, Homma Y, Pidet O, Guissou I, Hernigou J. Ceramic-on-ceramic bearing decreases the cumulative long-term risk of dislocation. Clin Orthop Relat Res. 2013;471:3875-82. [70] Hernigou P, Roussignol X, Delambre J, Poignard A, Flouzat-Lachaniette CH. Ceramic-on-ceramic THA Associated With Fewer Dislocations and Less Muscle Degeneration by Preserving Muscle Progenitors. Clin Orthop Relat Res. 2015. [71] Pitto RP, Garland M, Sedel L. Are Ceramic-on-ceramic Bearings in Total Hip Arthroplasty Associated With Reduced Revision Risk for Late Dislocation? Clin Orthop Relat Res. 2015. [72] Tsukada S, Wakui M. Lower Dislocation Rate Following Total Hip Arthroplasty via Direct Anterior Approach than via Posterior Approach: Five-Year-Average Follow-Up Results. The open orthopaedics journal. 2015;9:157-62. [73] Kurtz SM, Gawel HA, Patel JD. History and systematic review of wear and osteolysis outcomes for first-generation highly crosslinked polyethylene. Clin Orthop Relat Res. 2011;469:2262-77. [74] Sexton SA, Walter WL, Jackson MP, De Steiger R, Stanford T. Ceramic-on-ceramic bearing surface and risk of revision due to dislocation after primary total hip replacement. J Bone Joint Surg Br. 2009;91:1448-53. [75] Evangelista GT, Fulkerson E, Kummer F, Di Cesare PE. Surface damage to an Oxinium femoral head prosthesis after dislocation. J Bone Joint Surg Br. 2007;89:535-7. [76] Kop AM, Whitewood C, Johnston DJ. Damage of oxinium femoral heads subsequent to hip arthroplasty dislocation three retrieval case studies. J Arthroplasty. 2007;22:775-9. [77] Jaffe WL, Strauss EJ, Cardinale M, Herrera L, Kummer FJ. Surface oxidized zirconium total hip arthroplasty head damage due to closed reduction effects on polyethylene wear. J Arthroplasty. 2009;24:898-902. [78] Mai KT, Verioti C, D'Lima D, Colwell CW, Jr., Ezzet KA, Jr. Surface roughness of femoral head prostheses after dislocation. Am J Orthop (Belle Mead NJ). 2010;39:495-500. [79] McCalden RW, Charron KD, Davidson RD, Teeter MG, Holdsworth DW. Damage of an Oxinium femoral head and polyethylene liner following 'routine' total hip replacement. J Bone Joint Surg Br. 2011;93:409-13. [80] Gibon E, Scemama C, David B, Hamadouche M. Oxinium femoral head damage generated by a metallic foreign body within the polyethylene cup following recurrent dislocation episodes. Orthop Traumatol Surg Res. 2013;99:865-9. [81] Tribe H, Malek S, Stammers J, Ranawat V, Skinner JA. Advanced wear of an Oxinium femoral head implant following polyethylene liner dislocation. Ann R Coll Surg Engl. 2013;95:e133-5. [82] Moussa ME, Esposito CI, Elpers ME, Wright TM, Padgett DE. Hip dislocation increases roughness of oxidized zirconium femoral heads in total hip arthroplasty: an analysis of 59 retrievals. J Arthroplasty. 2015;30:713-7. [83] Garvin KL, Hartman CW, Mangla J, Murdoch N, Martell JM. Wear analysis in THA utilizing oxidized zirconium and crosslinked polyethylene. Clin Orthop Relat Res. 2009;467:141-5. [84] Lewis PM, Moore CA, Olsen M, Schemitsch EH, Waddell JP. Comparison of mid-term clinical outcomes after primary total hip arthroplasty with Oxinium vs cobalt chrome femoral heads. Orthopedics. 2008;31. [85] Morison ZA, Patil S, Khan HA, Bogoch ER, Schemitsch EH, Waddell JP. A randomized controlled trial comparing Oxinium and cobalt-chrome on standard and cross-linked polyethylene. J Arthroplasty. 2014;29:164-8. [86] Garvin KL, White TC, Dusad A, Hartman CW, Martell J. Low Wear Rates Seen in THAs With Highly Crosslinked Polyethylene at 9 to 14 Years in Patients Younger Than Age 50 Years. Clin Orthop Relat Res. 2015. [87] Jassim SS, Patel S, Wardle N, Tahmassebi J, Middleton R, Shardlow DL, Stephen A, Hutchinson J, Haddad FS. Five-year comparison of wear using oxidised zirconium and cobalt-chrome femoral heads in total hip arthroplasty: a multicentre randomised controlled trial. The bone & joint journal. 2015;97-B:883-9. [88] Zaoui A, Hage SE, Langlois J, Scemama C, Courpied JP, Hamadouche M. Do Oxidized Zirconium Femoral Heads Reduce Polyethylene Wear in Cemented THAs? A Blinded Randomized Clinical Trial. Clin Orthop Relat Res. 2015. [89] Heros R. Personal communication. CeramTec; 2015. [90] Dong YL, Li T, Xiao K, Bian YY, Weng XS. Ceramic on Ceramic or Ceramic-on-polyethylene for Total Hip Arthroplasty: A Systemic Review and Meta-analysis of Prospective Randomized Studies. Chin Med J (Engl). 2015;128:1223-31. [91] Si HB, Zeng Y, Cao F, Pei FX, Shen B. Is a ceramic-on-ceramic bearing really superior to ceramic-on-polyethylene for primary total hip arthroplasty? A systematic review and meta-analysis of randomised controlled trials. Hip Int. 2015:0.




